U.S. CDC Debates RSV Protection for Infants

The U.S. CDC's Advisory Committee on Immunization Practices (ACIP) is meeting today to review Respiratory Syncytial Virus (RSV) Maternal/Pediatric vaccine and a long-acting monoclonal antibody.
On August 3, 2023, Dr. Grace Lee is leading the ACIP meeting agenda, which includes, but is not limited to, the following presentations:
-
Introduction - Dr. S Long
-
EtR summary for nirsevimab - Dr. J Jones
-
Nirsevimab implementation considerations - Dr. G Peacock
-
Clinical considerations for nirsevimab & Workgroup considerations / proposed recommendations - Dr. J Jones
At around 2 pm ET today, the ACIP is scheduled to vote on two recommendations.
Previously, the U.S. Food and Drug Administration approved Beyfortus (nirsevimab-alip) for the prevention of RSV lower respiratory tract disease in neonates and infants born during or entering their first RSV season and in children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season.
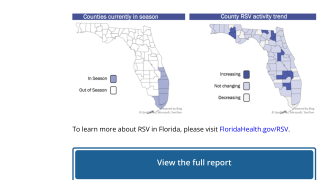
In the U.S., the RSV season generally starts in Florida in the fall. As of August 3, 2023, there have not been any RSV outbreaks reported this year.
The ACIP unanimously recommends routine use of Beyfortus™ to protect all infants below 8 months of age. The committee also voted unanimously to include Beyfortus in the Vaccines for Children program, supporting equitable access for all eligible infants.
Our Trust Standards: Medical Advisory Committee