Norovirus Vaccine Candidate Clinical Trials Advance
A clinical-stage biopharmaceutical company recently highlighted the progress and upcoming milestones for its investigational vaccine, HIL-214.
This vaccine aims to prevent moderate-to-severe acute gastroenteritis (AGE) caused by norovirus.
HilleVax, Inc. reported on March 20, 2024, topline data from the NEST-IN1 Phase 2B clinical study of HIL-214 in infants expected in mid-2024.
This is important news since no U.S. FDA vaccine is approved for AGE prevention. Noroviruses' genetic and antigenic diversity makes it difficult to develop a broadly effective vaccine.
According to the U.S. CDC, norovirus is a common intestinal infection marked by watery diarrhea, vomiting, abdominal cramps, nausea, and sometimes fever that may lead to clinically significant dehydration.
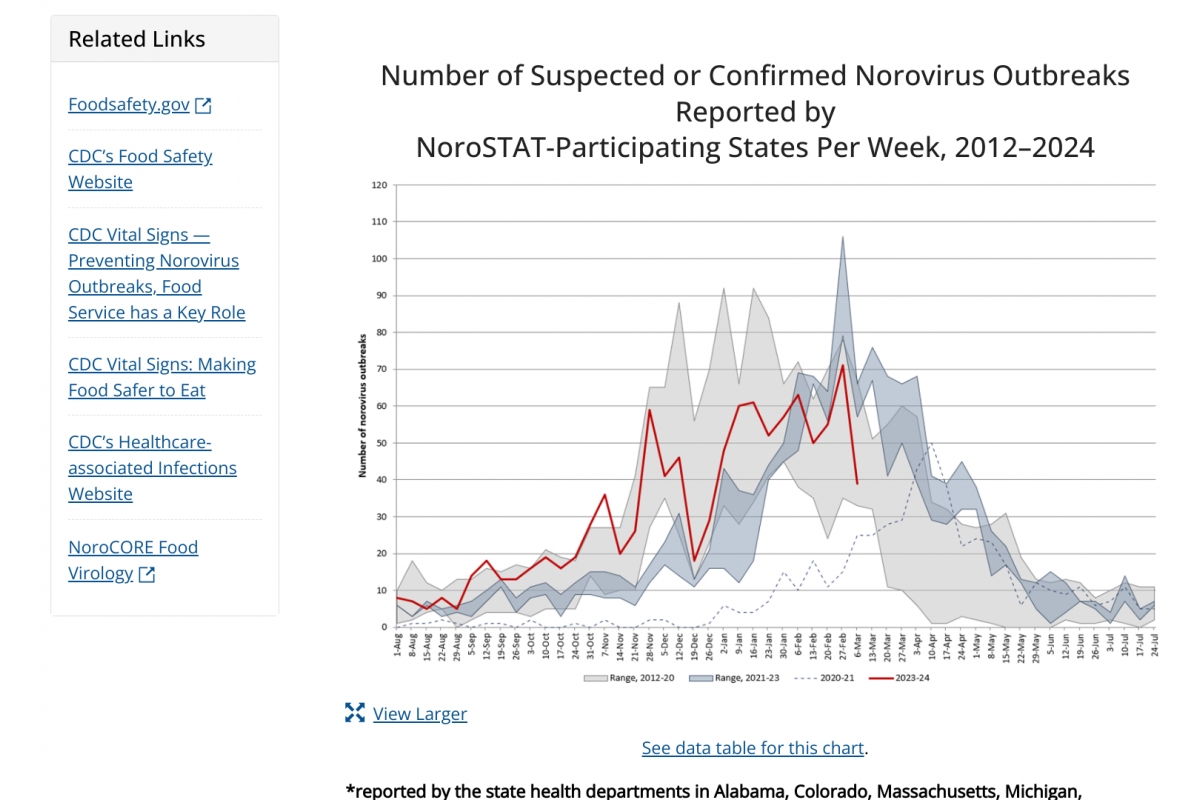
According to the CDC, 1,020 norovirus outbreaks were reported between August 2023 and March 12, 2024, within the 2012–2020 seasonal years range.
According to the CDC, three confirmed norovirus outbreaks on cruise ships were under U.S. jurisdiction in 2024.
"Over the past year, we made significant progress with HIL-214, our vaccine candidate for preventing moderate-to-severe norovirus-related AGE. We have continued to progress the NEST-IN1 clinical trial and remain on track to report topline data by mid-2024," said Rob Hershberg, MD, PhD, Chairman and Chief Executive Officer of HilleVax, in a press release.
"We have also made good progress on our manufacturing activities for HIL-214 in support of continued development in subsequent registrational trials for infants and older adults."
According to HilleVax, HIL-214 is a virus-like particle-based vaccine candidate.
In late 2023, HilleVax initiated three clinical trials supporting the advancement of HIL-214, including NOR-109, a Phase 1 clinical trial of HIL-214 in Japanese infants, NOR-206, a Phase 2 clinical trial co-administering HIL-214 with other standard infant vaccinations, and NOR-215, a Phase 2 serology study of HIL-214 in adults.
As of October 2023, HilleVax has completed enrollment of subjects in all three clinical trials.
Globally, norovirus is estimated to result in approximately 700 million cases of AGE and 200,000 deaths per year, resulting in direct and indirect healthcare system and societal costs of $10 billion in the U.S. and $60 billion globally.
Our Trust Standards: Medical Advisory Committee