Canada Approves Chikungunya Vaccine

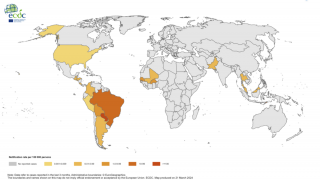
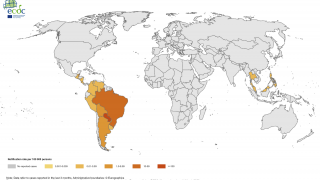
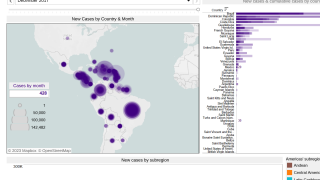
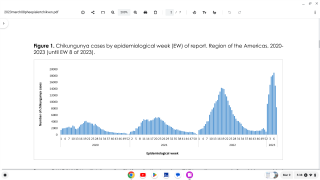
Since 2004, the Chikungunya virus (CHIKV) has caused large-scale outbreaks worldwide and has recently been identified in over 110 countries.
According to news from Valneva SE, a second country has approved IXCHIQ®, the world's first licensed chikungunya vaccine, to address this unmet medical need.
Valneva announced on June 24, 2024, that Health Canada had approved the IXCHIQ vaccine to prevent this mosquito-transmitted disease for adults.
This new approval follows the U.S. Food and Drug Administration decision in November 2023. The European Medicines Agency recently recommended marketing authorization for IXCHIQ in Europe, and a formal decision is expected in the third quarter of 2024.
On June 26, 2024, the U.S. CDC's vaccine committee plans to discuss how this innovative vaccine can be offered in the United States.
Chikungunya's economic burden is expected to increase as the mosquito vectors transmitting CHIKV spread geographically. As such, the World Health Organization recently highlighted chikungunya outbreaks as a significant public health problem.
Our Trust Standards: Medical Advisory Committee