Flu Shot Recommendations Updated for 2019-2020

Each year, influenza viruses typically circulate in the United States, most commonly from late fall through early spring.
Which is why the Centers for Disease Control and Prevention (CDC) strongly encourage everyone to get an annual flu shot.
But, the effectiveness of influenza vaccines varies depending on several factors, such as the types and subtypes of circulating influenza viruses, and the degree of similarity between circulating viruses and those included in the vaccine.
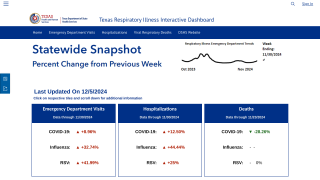
A new report published on August 23, 2019, updates the recommendations of the CDC’s Advisory Committee on Immunization Practices (ACIP) regarding the use of seasonal influenza vaccines in the United States for the 2019-2020 season.
The primary updates include the following 2 items:
- First, 2019–20 U.S. trivalent influenza vaccines will contain hemagglutinin (HA) derived from an A/Brisbane/02/2018 (H1N1)pdm09–like virus, an A/Kansas/14/2017 (H3N2)–like virus, and a B/Colorado/06/2017–like virus (Victoria lineage).
- And, quadrivalent influenza vaccines will contain HA derived from these 3 viruses, and a B/Phuket/3073/2013–like virus (Yamagata lineage).
- Second, recent labeling changes for two IIV4s, Afluria Quadrivalent and Fluzone Quadrivalent are discussed.
- The age indication for Afluria Quadrivalent has been expanded from ≥5 years to ≥6 months. The dose-volume for Afluria Quadrivalent is 0.25 mL for children aged 6 through 35 months and 0.5 mL for all persons aged ≥36 months (≥3 years).
- And, the dose-volume for Fluzone Quadrivalent for children aged 6 through 35 months, which was previously 0.25 mL, is now either 0.25 mL or 0.5 mL. The dose-volume for Fluzone Quadrivalent is 0.5 mL for all persons aged ≥36 months (≥3 years).
These ACIP recommendations apply to U.S.-licensed influenza vaccines used within the Food and Drug Administration–licensed indications.
These updates and other information are available from the CDC’s influenza website.
The ACIP provides annual recommendations for the use of influenza vaccines for the prevention and control of influenza. The ACIP Influenza Work Group meets by teleconference once to twice per month throughout the year. Additional information is available here.
Most pharmacies in the USA offer flu shot services beginning August each year. And, financial support programs for vaccines can be found at Vaccine Discounts.
Our Trust Standards: Medical Advisory Committee