Flu Shots Worked Better than Earlier Reports

New influenza vaccine effectiveness data presented at the U.S. CDC’s June 22, 2022 meeting of the Advisory Committee on Immunization Practices (ACIP) show flu shots worked better during 2021 - 2022 than initially reported.
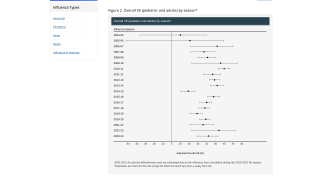
Published on June 23, 2022, this ACIP data shows flu shots reduced the risk of influenza illness by about 35% among vaccinated people.
Data from October 4, 2021, through April 30, 2022, showed that flu vaccines reduced people’s risk of mild to moderate flu illness caused by H3N2 flu viruses—the most common flu viruses this season—by about one-third overall.
The CDC’s early-season vaccine effectiveness estimates released in March 2022 were not statistically significant, suggesting vaccination did not offer benefits in reducing the risk of mild to moderate flu illness.
The early data underestimated how well flu vaccines worked during the 2021-2022 flu season because of the small sample size and biases due to COVID-19.
However, more data have since become available, and CDC has corrected for COVID-19 bias by excluding patients with COVID-19 illness from the comparison group.
These updated estimates are similar to what has been seen during past H3N2-predominant seasons and align with initial agency expectations based on laboratory data.
However, estimates of how well the flu vaccine protects against serious outcomes like hospitalization and ICU admission are pending.
Also, at the meeting, ACIP voted in favor of a preferential recommendation for certain flu vaccines over others for adults 65 years and older in the United States.
The ACIP voted to preferentially recommend higher-dose flu vaccines (Fluzone High-Dose vaccine and Flublok recombinant vaccine) or adjuvanted flu vaccine (Fluad vaccine) over standard-dose unadjuvanted flu vaccines.
And if one of these vaccines is unavailable at the time of administration, people in this age group should get a standard-dose flu vaccine instead.
This recommendation was based on a review of available studies, which suggests that, in this age group, these vaccines are potentially more effective than standard-dose unadjuvanted flu vaccines.
In recent years, CDC has not recommended any flu vaccine over another for any age group, and there is still no preferential recommendation for people younger than 65. However, CDC and ACIP will continue to review data on the comparative effectiveness of flu vaccines in different age groups as more information becomes available.
The CDC confirmed on June 22, 2022, that 2022–2023 Northern Hemisphere influenza vaccines would include updated reference viruses for A(H3N2) (clade 3C.2a1b, subclade 2a.2) and B/Victoria (B/Austria/1359417/2021).
More information on the U.S. vaccines with a preferential recommendation for older adults:
- Fluzone High-Dose Quadrivalent is an inactivated influenza vaccine for people 65 years and older. It contains four times the antigen, the part of the vaccine that helps the body build up protection against flu viruses, compared to standard-dose inactivated flu vaccines. The higher antigen dose is intended to give people 65 years and older a better immune response to vaccination and, therefore, better protection against flu.
- Fluad Quadrivalent is an adjuvanted inactivated influenza vaccine approved for use in people 65 years and older. It contains the same amount of antigen as other standard-dose inactivated flu vaccines plus MF59 adjuvant. The adjuvant is intended to give people 65 years and older a better immune response to vaccination and, therefore, better protection against flu.
- Flublok Quadrivalent is a recombinant protein influenza vaccine for people 18 years and older. It is made using different production technology than the inactivated influenza vaccines and contains three times the antigen dose compared with standard-dose inactivated flu vaccines. The higher antigen dose is intended to give people 65 years and older a better immune response to vaccination and, therefore, better protection against flu.
The U.S. CDC published updated influenza vaccine schedules for 2022 on Feb. 17, 2022. The CDC continues to suggest that everyone six months and older should get an annual flu vaccine to protect against flu and its potentially serious complications.
This is especially true for people at higher risk of developing serious flu complications, including people 65 years and older, pregnant people, young children, and people with certain chronic medical conditions.
Unfortunately, flu vaccination coverage for children six months to 17 years has been about 2% points lower this season as compared with last season (55.3% compared to 57.3%)
If you have questions about which influenza vaccine is best for you, talk to your doctor, pharmacist, or other healthcare professional.
Additional influenza vaccine news is posted at PrecisionVaccinations.com/Flu.
PrecisionVaccinations publishes fact-checked, research-based vaccine news curated for mobile readership.
Our Trust Standards: Medical Advisory Committee