Guinea Ebola Outbreak Declared Over

The Ebola outbreak that emerged in Guinea in February 2021 has been declared over by the World Health Organization (WHO) in Africa.
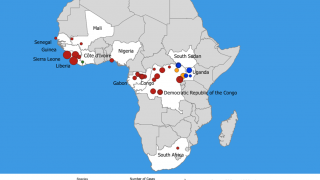
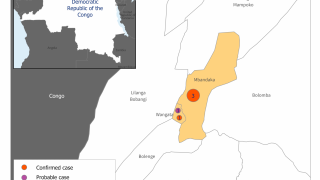
A total of 16 confirmed and seven probable cases were reported in Guinea’s latest outbreak. Eleven patients survived, and 12 lives were lost.
Guinean health authorities declared this Ebola outbreak in February 2021 after three cases were detected in the southern N’zerekore prefecture. This is the same region where the 2014–2016 Ebola outbreak emerged before spreading into neighboring Liberia and Sierra Leone.
Though more studies are needed to understand how the two outbreaks may be linked fully, Guinean health authorities reactivated a surveillance program for survivors to provide long-term monitoring and after-care support, says the WHO.
The WHO helped ship around 24,000 Ervebo vaccine doses and supported the vaccination of nearly 11,000 people at high risk, including over 2,800 frontline workers.
Merck’s Ervebo, Ebola Zaire Vaccine, Live, formerly known as V920 (rVSV-ZEBOV-GP), is a recombinant, replication-competent Ebola vaccine. Ervebo's active ingredient is live Vesicular Stomatitis Virus, in which its surface protein has been replaced with that of Zaire ebola virus disease.
Dr. Matshidiso Moeti, WHO Regional Director for Africa, stated in a press statement issued on June 17, 2021, “We are getting faster, better, and smarter at fighting Ebola. But while this outbreak is over, we must stay alert for a possible resurgence and ensure the expertise in Ebola expands to other health threats such as COVID-19.”
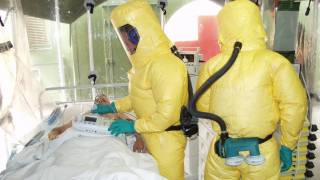
An Ebola laboratory, treatment infrastructure, logistics capacity, and infection prevention measures have been reinforced in Guinea to better respond to the disease and other health emergencies.
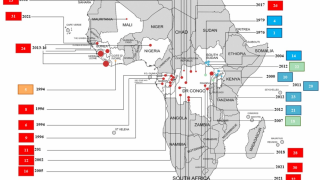
While the latest Ebola outbreak was limited to Guinea, to prevent cross-border infections, WHO supported six of the country’s neighbor countries to ramp up preparedness measures, including stepping up surveillance and screening at border crossings and within high-risk communities, as well as enhancing coordination between governments and the respective health services.
In support of the government’s efforts to curb the outbreak, WHO worked with other United Nations agencies and partners such as the African Development Bank, Alliance for International Medical Action, African Centres for Disease Control and Prevention, European Civil Protection and Humanitarian Aid Operations, Gavi, the Vaccine Alliance, the International Organization for Migration, Global Outbreak Alert and Response Network, Terre des Hommes, United Nations Central Emergency Response Fund, United Nations Children’s Fund, United States Agency for International Development, World Bank and World Food Programme.
Vax-Before-Travel publishes research-based vaccine news.
Our Trust Standards: Medical Advisory Committee