India’s Zika Virus Concerns Continue

The World Health Organization (WHO) confirmed in early October 2021, a Zika virus (ZIKV) infection was laboratory-confirmed in a resident of Kerala state, south-west India.
This 24-year-old pregnant woman in her third trimester of pregnancy represents the first Zika virus disease case ever reported from Kerala.
On June 28, 2021, she was admitted to a private hospital with arbovirus like symptoms of fever, headache, and general rash. Laboratory results were negative for dengue virus (DENV) and chikungunya virus (CHIKV).
The patient delivered her child in early July, and there were no apparent congenital disabilities in the newborn.
Her mother reported having fever and similar symptoms one week before ZIKV confirmation in her daughter among her close contacts.
Retrospective testing was conducted among 19 hospital staff and patients at the same private hospital who had previously presented with fever, myalgia, arthralgia, and petechial lesions in May 2021.
Blood samples collected from these 19 ZIKV suspected cases were sent to NIV Pune. On 10 July, the laboratory results confirmed that 13 of the 19 samples tested positive for ZIKV by RT-PCR, indicating cryptic transmission of ZIKV in Kerala state since May 2021.
From 8 to 26 July 2021, 590 blood samples were collected in Kerala state through active case finding and passive surveillance.
Of tests, 70 (11.9%) were positive for ZIKV by RT-PCR at NIV Pune, including four additional pregnant women.
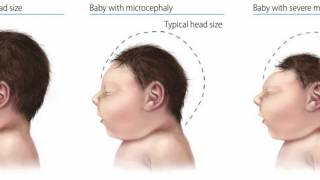
So far, no cases of microcephaly and/or Guillain-Barre syndrome (GBS) have been linked with this Zika outbreak.
In response, the Kerala Health department, along with local self-government health departments, has implemented the following response activities:
- On 8 July 2021, the State of Kerala issued guidelines on enhanced surveillance for ZIKV disease and sent guidance to all 14 districts.
- Information, Education, and Communication activities about ZIKV disease have been strengthened immediately throughout the State. In addition, sensitization activities across the State for both health care workers and the general public are ongoing.
- All ultrasound scanning centers have been directed to report incidences of microcephaly during regular antenatal scans to the Reproductive and Child Health Officer.
- Currently, four laboratories (National Institute of Virology Alappuzha, Medical College laboratories of Trivandrum, Thrissur, and Kozhikode) in Kerala state are equipped to perform RT-PCR testing for ZIKV infection. In addition, the State is also planning to start testing in another public health laboratory in the Trivandrum district. So far, the State has received 2100 RT-PCR kits from NIV Pune to detect Zika cases, distributed to the four laboratories mentioned above.
- Measures to ensure strict deferral of blood donors with a history of fever in the previous two weeks have been undertaken.
- A central team visited the Trivandrum district, collected samples of mosquitoes and larvae from the residential area of the cases, and sent them for testing at the Vector Control Research center, Kottayam field station in Kerala state. The results are pending.
- The State Health Minister has conducted multiple rounds of reviews. As a result, all the districts have been alerted to carry out active surveillance, mosquito control and information, education, and communication activities related to the management of ZIKV.
- In the Trivandrum district, which has been declared a cluster of ZIKV disease cases, intensified vector control activities have been conducted for a week, including; extensive fogging, spraying, larvicides, source reduction, and sanitization of the surrounding areas. Additionally, field teams visited each household to conduct active case findings, ensure elimination of mosquito breeding sites, and sensitize the community to preventive mosquito control measures and identification of ZIKV disease symptoms to seek timely medical assistance.
The WHO was requested to support the enhanced surveillance among antenatal women, microcephaly surveillance, and Acute Flaccid Paralysis (AFP) and GBS.
The WHO says ZIKV can cause large epidemics with a substantial demand on the public health system, including surveillance, case management, and laboratory capacity to differentiate ZIKV disease from illness due to co-circulating mosquito-borne viruses like dengue and chikungunya.
About 60-80% of the Zika virus infected cases are asymptomatic or have mild symptoms; ZIKV can cause microcephaly, congenital Zika syndrome, and GBS.
Moreover, although Aedes species mosquitoes primarily transmit ZIKV, it can also be transmitted from mother to fetus during pregnancy through sexual contact, transfusion of blood and blood products, and organ transplantation.
Until protective vaccines become available, the WHO says protection against mosquito bites during the day and early evening is crucial for preventing ZIKV infection. And special attention should be given to avoiding mosquito bites among pregnant women, women of reproductive age, and young children.
For regions with active transmission of ZIKV, all persons with suspected ZIKV infection and their sexual partners (particularly pregnant women) should receive information about the risks of sexual transmission of ZIKV.
The WHO recommends that sexually active men and women be correctly counseled about ZIKV infection and offers a full range of contraceptive methods to decide whether and when to become pregnant to prevent congenital Zika syndrome other possible adverse fetal pregnancy outcomes.
And pregnant women should practice safer sex (including correct and consistent use of condoms) or abstain from sexual activity for the entire duration of pregnancy.
In addition, following the state/national response plan, pregnant women should be encouraged to attend scheduled appointments and enhance antenatal care and follow-up, including ultrasound imaging to detect microcephaly and other developmental anomalies associated with ZIKV infection in pregnancy, following the state/national response plan.
A listing of Zika virus vaccine candidates is published on this webpage.
ZikaNews publishes fact-check, research-based Zika vaccine news.
Our Trust Standards: Medical Advisory Committee