$120 Million Financing to Advance MAPS Vaccines

A Massachusetts based biotechnology company announced the completion of a $120 million Series B financing round.
Announced on April 23, 2020, Affinivax Inc. said in a press release it is advancing its novel Multiple Antigen Presentation System (“MAPS”) vaccine technology into clinical trials.
Steven B. Brugger, CEO of Affinivax, “We are very excited to look ahead to our next phase of growth, taking our lead pneumococcal vaccine through Phase 3 testing and bringing several additional novel MAPS vaccines and immunotherapies into clinical trials.”
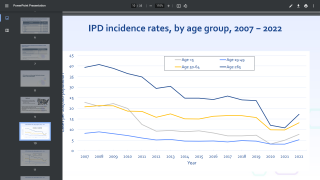
Affinivax’s lead vaccine targeting Streptococcus pneumoniae (ASP3772, partnered with Astellas Pharma) includes 24 pneumococcal serotypes, more than any other vaccine on the market or in clinical testing today.
ASP3772 is currently being studied in pneumococcal infection in elderly adults, its initial indication, in an ongoing Phase 2 study that is nearing completion.
In a previously completed Phase 1 study, ASP3772 was demonstrated to be safe and highly immunogenic in healthy adults. The clinical program for ASP3772 is expected to expand to include an infant indication.
Affinivax recognizes the ongoing need to increase protection against new, emerging pneumococcal serotypes and will continue to focus on developing additional pneumococcal MAPS vaccines designed to prevent against an even greater number of serotypes.
The MAPS technology platform uses proprietary chemistry that capitalizes on the highly specific and durable non-covalent, affinity binding between biotin and rhizavidin, a biotin-binding protein.
The highly stable MAPS complex created by this affinity binding contributes to a simple, modular, and efficient approach to the development of novel vaccines and immunotherapies.
Conventional vaccine conjugation technology seeks to optimize the generation of protective antibody responses mainly to polysaccharide antigens, using the protein antigen as a carrier.
In stark contrast, a MAPS vaccine can present both the polysaccharide and the protein antigens to the host immune system to induce both a B- and T-cell immune response.
This unique capability of the MAPS technology allows for the tailored development of each MAPS vaccine or immunotherapy based on the specific type of immune response that is most desired for each pathogen and disease.
Affinivax is advancing a next-generation technology platform to enable the development of vaccines and immunotherapies.
Pneumococcal vaccine development news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee