Should Florida Vaccinate Children Against Dengue

One of the world's most complex mosquito-borne viral diseases is making a more significant impact in the U.S. this year. Annually, Dengue is transmitted to about 100 million humans through the bite of infected mosquitoes.
Before 1970, only nine countries had experienced severe dengue epidemics, says the World Health Organization.
Since then, Dengue has become endemic in Puerto Rico, American Samoa, the U.S. Virgin Islands, the Federated States of Micronesia, the Republic of Marshall Islands, and the Republic of Palau.
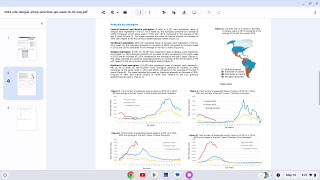
From 2010 to 2020, approximately 95% of locally acquired dengue cases in the U.S. occurred in Puerto Rico (n = 29,779).
And during 2022, Puerto Rico reported over 300 locally-acquired dengue cases.
Separately, Florida has also confirmed 582 travel-related and 32 locally-acquired dengue cases year-to-date.
Most of Florida's local cases were reported in the Miami area last summer.
The good news is that a U.S. FDA-approved preventive vaccine is available. Dengvaxia is a live attenuated tetravalent chimeric vaccine made using recombinant DNA technology.
During the last U.S. CDC Advisory Committee on Immunization Practices (ACIP) meeting in October 2022, Wilbur Chen, MD, MSc, led a presentation focused on the steps Puerto Rico is taking to protect adolescents from Dengue with an extensive vaccination program.
In Puerto Rico, about 280,000 adolescents 9–16 years old might be eligible for Dengvaxia.
Multiple visits to healthcare providers and/or the laboratory are required to determine eligibility for Dengvaxia and start the vaccination series.
The program launched on September 7, 2022, when Dengvaxia was administered to the initial children in Puerto Rico.
The three-phase implementation plan in Puerto Rico will take several months, focused on vaccination safety.
Upon completion, the program could be a model for Florida to emulate.
If the Dengvaxia protocol is too difficult, there is a new dengue vaccine the U.S. CDC is reviewing.
The Qdenga® vaccine was developed and is significant because it is the first vaccine for people who have not been exposed to Dengue.
The Indonesian drug regulator has already approved its use without testing for previous exposure in August. And Europe's drug regulator (EMA) is also considering approving the vaccine without testing.
Should Florida launch a vaccination program, it may be able to select either Dengue vaccine.
Vax-Before-Travel publishes fact-checked, research-based travel vaccine news manually curated for mobile readers.
Our Trust Standards: Medical Advisory Committee